Cognitive plasticity
How life becomes agential
Agency has been the focus of many Big Biology episodes, so much so that I decided to gather my thoughts on the topic. In particular, I thought it would be useful to merge points made by previous guests (and many other thinkers!) with related things I’m studying in my research lab. Below is a synopsis of those ideas. Apologies that the text reads more like a journal article than a blog post, but I hope you find something valuable here! Marty
Abstract: A key concept for understanding and measuring phenotypic plasticity is the reaction norm, a method that emerged in the early 20th century and has since become an important tool for biologists. The power of the reaction norm derives from partitioning sources of phenotypic variation into genetic (G), environmental (E), and interactive (GxE) causes, which has facilitated major advances in our understanding of phenotypic complexity in ecology and evolution. The simplifying power of the reaction norm, however, could also be a weakness– we now understand that many different kinds of information beyond genes influence living systems, so decomposition of trait variation into G, E and GxE will often be insufficient. Like genomes, environments are complex, and we often do not know what about them organisms are sensing and responding to. Recent progress in systems biology underscores another weakness of the reaction norm mindset: that organisms consist of interlocking sets of processes, distributed across levels of biological organization, each with extraordinary capacities for learning and refining decisions based on experiences. Here, I propose that the concept of cognitive plasticity captures these issues well. By adopting cognition as a central concept for certain forms of plasticity, we could produce a better framework for understanding the causes and consequences of phenotypic variation within and among generations, perhaps eventually linking the smallest to the largest levels of biological organization.
Natural selection is not a consequence of how well the organism solves a set of fixed problems posed by the environment; on the contrary, the environment and the organisms actively codetermine each other. (Lewontin 1970)
An organism does not just encode a model of the world; it is a model of the world—a physical transcription of causal regularities in its eco-niche that has been sculpted by reciprocal interactions between self-organization and selection over time. (Ramstead et al. 2018)
Learning is optimization by trial and error, and so is evolution. (Vanchurin et al. 2022)
Plasticity as a core biological process
Phenotypic plasticity—the ability of living systems to alter their phenotypes in response to changing environmental conditions—is central to biology, and the main analytical framework for understanding plasticity is the reaction norm (Schlichting and Pigliucci 1998). In reaction norms, one quantitatively ascribes phenotypic variation to either genetic (G) or environmental (E) sources, or to their interaction (GxE) (Via et al. 1995). This approach has yielded significant advances, from the development of sophisticated methods for estimating these forces from increasingly complex datasets (Nussey et al. 2007, Lande 2009, Dingemanse et al. 2010, Martin et al. 2011, Smallegange 2022, Dupont et al. 2024) to inference about the trajectories of populations into the future (Jablonka et al. 2014, West-Eberhard 2003). Despite these successes, reaction norms might have sometimes misled us, at least when it comes to a very common sets of plastic traits, namely the cognitive ones. Cognitively plastic traits are not traditionally plastic traits at all; they are purposive processes (Nicholson and Dupré 2018). Whereas this statement might seem contentious because of ‘purposive’ has spooky connotations, it means that to understand some if not most biological traits, mechanistically but also evolutionarily, we would probably benefit from a different approach than the one offered by the reaction norm.
Here, like others, I define cognitive plasticity as the subset of plastic phenotypic traits arising from the dynamic interplay of information from the environment (Adami 2024) with a living system’s existing model of its world, including but not limited to its genes (Levin and Dennett 2020, Vanchurin et al. 2022, Watson and Szathmáry 2016). Cognitive plasticity thus emphasizes that many plastic traits are functional and of evolutionary consequence because they become differently adaptive through time. Ideas like this one have been proposed before (Baldwin 2018, Gottlieb 2002), but for the most part, this perspective is distinct from the dominant framework for understanding phenotypic variation and evolutionary change, the Modern Synthesis (Laland et al. 2014, Huxley 1942). The Modern Synthesis proposed that phenotypic diversity, including adaptation, is the outcome of genetic variation interacting with selection across generations. While broadly valid, this portrayal is very generic, and indeed does not capture the fundamental features of life (Walsh 2015, Laland et al. 2014, Feiner et al. 2024), that living systems are entangled sets of information-rich processes that exist far from thermodynamic equilibrium. The Modern Synthesis’ strong focus on genes has been very productive in the practical sense, but it has also led us, sometimes unintentionally, to ascribe to genes a causal power that exceeds their actual roles (Pigliucci 2007, Pigliucci 2010, Noble 2008). I think that this position is no longer defensible because as Sewall Wright said (Wright 1931) and Walsh and Sultan (2024) have recently reminded us that individual adaptability, not genes, is probably the chief object of selection.
Phenotypic plasticity and the shortcomings of reaction norms
Over the past century, many biologists have that emphasized that phenotypic plasticity is a phenomenon central to evolution (Schmalhausen 1949, Baldwin 2018, Waddington 1953). Plasticity is so compelling and so powerful because it highlights the dual role of the environment in evolution (West-Eberhard 2003): environments both induce and select phenotypic variation. These dual roles have led to excitement and conflict (Walsh 2015, Walsh and Sultan 2024, Laland et al. 2014). Some experts have suggested that plastic traits operate and evolve just like all others (Laland et al. 2014), and some have said that the existence of plasticity means that genes will more often be followers than leaders in evolution (West-Eberhard 2003, Waddington 1953). Others have claimed that plasticity (among other phenomena such as niche construction (Odling-Smee et al. 1996)) requires a complete reworking of our fundamental models of evolution (Moczek et al. 2011, Pigliucci and Müller 2010, Noble 2006, Jablonka et al. 2014, Feiner et al. 2024). Still, it is widely agreed that even simple forms of phenotypic plasticity can have major evolutionary impacts: they can shield genetic variation from selection (Price et al. 2003), release cryptic genetic variation in stressed populations (Ghalambor et al. 2007a), lead to genetic assimilation (Ledon-Rettig et al. 2010), and alter evolutionary patterns throughout whole genomes (Wade and Sultan 2023). In sum, plasticity is viewed a central to most realms of biology, but the question endures, is all plasticity understandable in the same way?
To date, most progress in the study of plasticity has benefited at least in part from the reaction norm construct, first proposed in 1909 (Woltereck 1909). Reaction norms function at two levels. First, they simply describe how traits of individuals or genotypes covary with aspects of the environment. Second, they provide a way to attribute (partition) variation in trait values to genetics (G), environments (E), their interaction (G x E), or much more complicated parsing (IxE, Nussey; GxExE, Kingsolver et al. 2006). Over the past several decades, better and better reaction norm models (e.g., random slope regression, random regression animal models, double-hierarchical GLMMs, etc. (Dingemanse et al. 2010, Dingemanse and Wolf 2013, Martin et al. 2011)) have been developed to understand many forms of plasticity (Araya-Ajoy and Dingemanse 2014), including those that parse the relative influence of genetic versus environmental effects experienced during development (Nussey et al. 2007, Westneat et al. 2011). Some reaction norm models have gone so far as to address how multiple environmental factors simultaneously affect plastic variation in one trait (Westneat et al. 2019), and others have been developed to describe plastic traits that account for some past experience (Wright et al. 2022). These methods work both for traits that vary continuously (e.g., height, metabolic rates) and traits that vary in more discrete ways (e.g., presence or absence of spots, spines, or scales), and they have been used to study plasticity at several levels of organization, from morphology and behavior (Snell-Rood 2013, Svensson et al. 2020, Martin et al. 2021, Hau et al. 2022) to physiology and even gene expression (Ghalambor et al. 2015, Rivera et al. 2021).
Despite all of this success, there are two issues that suggest our concept of plasticity to date has been too expansive. First, ‘plasticity’ has different meanings and different points of emphasis to different kinds of biologists. Semantic differences alone would not constitute a problem, but the plasticity studied by neuroscientists, immunologists, endocrinologists, and molecular biologists is not the plasticity that will yield to reaction norm style investigations. Partly, this is because whereas suborganismal biologists would often call neurogenesis, hormone regulation, and lymphocyte receptor development plastic, they rarely would parse variation in these traits into G, E and GxE (Martin et al. 2021). With exception, suborganismal biologists are either not interested in the evolutionary adaptiveness what they measure (Gluckman et al. 2009, Trotter et al. 2011), or they infer that regulated, plastic variation in neurogenesis or lymphocyte receptor diversity must be adaptive because there are so many ways such complex system could fail, how else could we explain these context-dependent, dynamic responses but via fitness (Haig 1996)? Occasionally, suborganismal researchers directly test for adaptiveness of plastic trait variation by examining individual differences in hormone levels or immune responses relative to fitness (Bonier et al. 2009, Arnold 1983), but mostly, biologists studying these forms of plasticity call their traits plastic just because they remain labile throughout the life of an individual. Who knows and who often cares how these traits fit into reaction norms?
The second issue about the expansive of the word plasticity is also highlighted by suborganismal biology. In particular, some plastic traits are dynamically plastic. Certain dynamic plasticities are simply reversible; an environment induces one phenotypic variant (i.e., a change in coat color), which then reverts to the original state prior to the environmental experience. Other plastic responses, though, never revert to the original condition; they enduringly change over the life of an individual, in many cases becoming functional because every new instantiation is adaptive and distinct from its historical form. Over the last decades, evolutionary biologists have tended to focus on comparatively simple forms of plasticity (Gilbert and Epel 2009, Feiner et al. 2024, Moczek et al. 2011, Ledon-Rettig et al. 2010). On the positive side, this approach enabled them to discern when plasticity was heritable, adaptive, non-adaptive or even maladaptive (Ghalambor et al. 2007b, Martin et al. 2021). On the downside, that success seems to have inspired an over-confidence that all plasticity is alike. In some cases, this perspective could hold. Reversible plasticities (e.g., seasonal changes in coloration or reproductive capacity) might well be decomposable with reaction norms (Piersma and Drent 2003, Williams 2008, Swanson and Merkord 2013, McWilliams and Karasov 2014), but traits that increase and decrease to different magnitudes and over very different timescales (Zimmer et al. 2022, Woods and Wilson 2014, Dantzer 2023, Dupont et al. 2024), often but not always returning to some baseline values, probably won’t. Unlike the timing of breeding, molting, migration or other reversible plasticities (Nelson et al. 2002), molecular epigenetic changes to genomes, metabolic rates, cell-cell interactions and all sorts of other suborganismal processes will never return to an original condition once elicited. Indeed, it is sometimes because they do not revert to earlier forms that they foster fitness (Sultan 2019).
These especially complex forms of plasticity probably evolved to vary among several forms throughout the life of an individual in a recurrently contingent way (Snell-Rood 2013, Snell-Rood and Ehlman 2021). This scenario also means that plastic variation in these traits will not typically be related to fitness (or even organismal function) in a simple way (Kingsolver et al. 2001). For plastic traits where the ‘solutions’ (i.e., fit of phenotype to environment) are very complex (e.g., infections, social interactions, invasion leading to new trophic interactions) or evolutionarily novel (e.g., during invasions or range expansions), cognitive plasticity will probably be necessary (Walsh and Sultan 2024, Martin and Zimmer 2022, Zimmer et al. 2022). Only through the screening, processing and use of (semiotic) information will a living system instantiate the trait variant that will solve the problem (Zimmer et al. 2022, O’Connor et al. 2019). Existing reaction norm methods simply cannot accommodate these situations, or at least they have not done so yet. Reaction norms, mostly, conceive plastic traits as static, simple things, but cognitive plasticities, traits that change or even improve system performance through time (Taleb 2014, Walsh and Sultan 2024), are deliberative processes that probably require a different paradigm (Nicholson 2019, Nicholson and Dupré 2018).
To illustrate this problem explicitly, consider a vertebrate adaptive immune response (Hedrick 2004). Hosts must mobilize a variety of defenses against each threat, then learn effectively from past experiences to protect themselves better from exposures in the future. One could, in principle, apply traditional reaction norm methods to many forms of immune data, parsing for instance how genetic, individual, and experiential factors contribute to variability in T and B cell responses (Martin et al. 2021). The equations cannot distinguish meaningful from suspect traits; any trait is potentially decomposable with a reaction norm. Even still, such studies would require immense sample sizes (Martin et al. 2011), making them typically underpowered or restricted to simpler organisms, like unicellular life. More significantly from the perspective of cognitive plasticity, describing reaction norms for arbitrarily selected immune traits would not necessarily capture the specific learning component inherent in adaptive immunity, the thing that makes it interesting to understand in the first place. Unlike other plastic traits, adaptive immune responses are antifragile (Taleb 2014); they improve in response to adversity and experience. Describing a reaction norm for antibody concentrations or some other adaptive immune trait would thus not capture the most important thing about adaptive immune plasticity we seek to know: whether and how the second response is more conducive to fitness than the first. Even sophisticated reaction norm approaches (Wright et al. 2022) will struggle with this kind of plasticity, as they have no means capture how subsequent forms of variation come to better fit a living system to a particular condition through time. Importantly, too, this situation does not apply solely to vertebrates. Invertebrates have functionally similar (i.e., cognitive) immune responses, just not the lymphocytes that produce antibodies and educated T cells (Kurtz and Armitage 2006). Most importantly, this rationale for immune responses applies to many if not most cognitively plastic traits, on any level of biological organization.
Defining and defending cognitive plasticity
Cognitive plasticity describes the regulatory capacity of an individual to mobilize a particular plastic trait variant through time. Importantly, mobilization changes the propensity of the individual to mitigate a challenge or exploit an affordance. The first step toward understanding these forms of plasticity, particularly their evolution, is to embrace that life is processual, not static as now is the prevailing fashion. As Woese wrote (Woese 2004), “it is becoming increasingly clear that to understand living systems in any deep sense, we must come to see them not materialistically, as machines, but as (stable) complex, dynamic organization.” Woese meant that living systems are alive because they are open physical systems that constantly exchange energy, matter, and information with their surroundings (Walker et al. 2016, Walker et al. 2017, Davies 2019). These traits help them maintain themselves far from thermodynamic equilibrium and thus distinguish living from other competent but non-living systems (Levin 2023, Turner 2016, Turner 2017).
Reaction norms, largely, encourage us to view living systems more like passive machines than entangled sets of informed processes (Nicholson 2019, Walker et al. 2017). Machines persist even without exchanging energy and matter with their surroundings -- think of your computer powered down and sitting in a backpack. Living systems exhibit dynamic stability (Davies 2019), meaning that you cannot power down your cat in the morning and expect to power it back up when you get home. Many things about living systems will therefore not yield to simple decomposition. Thinkers (Von Bertalanffy 1952, Waddington 2014) since Heraclitus have recognized that life is more like a stream than machine, but their ideas have yet to alter prevailing theory. As Nicholson (2019) writes,
The external form of a stream is stable only because of the constant flow of water molecules that enter into it and emerge out of it. The moment this flow is interrupted, the stream itself disappears, as its very existence depends on the steady movement of water passing through it. In the same way, the physical form of an organism is merely the visible expression of the constancy of catabolic and anabolic processes going on within it. Its persistence through time is entirely dependent on the extremely intricate balancing of these two opposing kinds of reactions.
The upshot: reaction norms imply life as decomposable and therefore static, but it is not. We therefore need to adjust our frameworks for understanding phenotypic variation, considering specifically reciprocal causality among genes, environments, their interactions (Mitchell 2023, Davies 2019, Svensson 2018) and the higher-order interactions they entail (Okasha 2006, Noble 2006).
The second step to studying cognitive plasticity effectively is to accept that many biological processes are purposive (Moczek and Sultan 2023, Walsh 2015, Turner 2017). Of course I don't mean purposive in the mystical manner that intelligent design advocates do. I mean purposive in the sense that living systems are information-rich systems with goals. Unlike simple forms of plasticity, which can be triggered by single or simple signals (Uvarov 1921), cognitive forms of plasticity emerge from the interplay of newly acquired information instantiated in comparatively transient substrates (i.e., neural, physiological, epigenetic) with older forms of information, genetic but also epigenetic and structural (Hofstadter 2007). In this light, cognitive plasticities will not involve the unspooling of genetic information into environmental space; they will be the outcomes of a set of integrated, deliberative processes, engaged to accomplish particular goals (Levin and Dennett 2020). Ironically, we have little trouble appreciating the uniqueness of cognitive plasticity in some contexts (Jablonka et al. 2014), namely when talking about actions that entail brains (Okasha 2018). Once we take more seriously that most all organisms are composed of nested, cognitive subsystems (Lyon et al. 2021, Levin 2023), a focus on cognitive plasticity becomes almost intuitive.
Here, it is critical to emphasize too, as François Jacob and Jacques Monod did long ago (Jacob and Monod 1961), that even simple forms of life are cognitive and thus make decisions. Systems need not even have brains to be cognitive. Escherichia coli, for instance, take in and metabolize glucose over other substrates when glucose is available. If glucose is absent but lactose is present, E. coli retools itself to produce the proteins needed for using lactose (Müller-Hill and Oehler 1996). How does a bacterium decide whether to switch? In the language of cognition, it understands relative concentrations of locally available sugars by making a protein (i.e., a transcription repressor) that binds reversibly to lactose. In E. coli, much like in human neurocognitive understanding, then, deciding involves an information filter (i.e., a means to distinguish signal from noise), subsequent processing, and eventual action (Wade and Sultan 2023). Bacteria thus have to discriminate meaningful (semiotic) information (i.e., the presence or absence of lactose) from the flood of other molecules in the environment (i.e., noise), and their informational filters (i.e., the lactose-binding repressor) enable them to do this (O’Connor et al. 2019). Once a resource is found, the cell decides whether it will switch its metabolism by linking the repressor’s lactose binding state to its repressing activity via use of its internal model (see below). If lactose levels in the cell rise high enough (and there is little or no glucose present), the repressor ceases to bind the operator, allowing transcription of the lac operon to proceed. The lac operon encodes three additional proteins: an intracellular enzyme that cleaves lactose into its components, a symporter that localizes to the membrane and renders it more permeable to lactose, and another enzyme that helps process monosaccharides internally. Ultimately, these phenomena sculpt each E. coli into a better model of its environment over its lifetime.
As a brief aside, cognitive plasticity appears to occur at even at the genomic level (Walsh 2015, Okasha 2018). Genomes actively undergo various modifications, such as cutting, transposing, and repairing DNA, and research since the 1930s indicates that expressed genetic variation emerges from regulated cellular processes rather than accidental mutations (Shapiro 2011).
A conceptual roadmap to studying cognitive plasticity
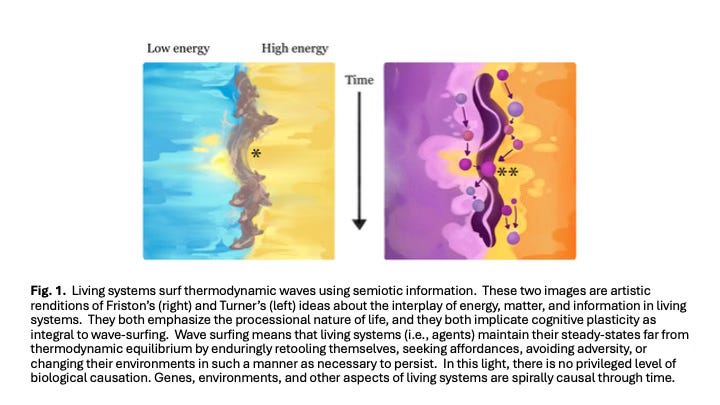
A major obstacle to understanding cognitive plasticity is the lack of a framework linking flows of matter, energy, and information (Hesp et al. 2019, Davies 2019). Some researchers suggest that a framework might emerge by focusing on the similarity between maximizing organismal performance within a lifetime and increasing population fitness over evolutionary time (Okasha 2018, Watson and Szathmáry 2016). Others propose frameworks built around integrated feed-forward and feedback loops (Del Giudice et al. 2018, Bernhardt et al. 2020). I agree with both perspectives and identify Karl Friston (Friston 2010) and J. Scott Turner (Turner 2016, Turner 2017) as thinkers whose proposals might significantly advance this discussion (Fig. 1).
According to Friston, cognition does not require a nervous system; entities exhibit cognition when their current or past experiences shape their future responses (Friston et al. 2023). The Free Energy Principle (FEP) posits that all non-equilibrium steady-state systems maintain viability through internal models that interpret and use environmental information received via a Markov blanket (Kirchhoff et al. 2018). In FEP, free energy quantifies uncertainty or surprise about the environment, reflecting the accuracy and confidence of a system’s predictions. Higher free energy corresponds to greater surprise. Minimizing variational free energy equates to minimizing thermodynamic entropy (Ramstead et al. 2018), enabling continuous model refinement (i.e., cognition and learning). Entities thus remain alive by predicting changes in internal and external conditions (Bayesian updating; Fig. 1A), then taking action to prevent entropic breakdown. Systems optimizing these processes achieve competitive and reproductive advantages. Although FEP is not without its skeptics (Colombo and Wright 2021), it provides a powerful way to understand cognition across diverse complex systems and levels of organization.
Turner’s approach (Turner 2016) defines free energy more traditionally—as energy available to perform work. He uses the metaphor of a standing thermodynamic wave (Fig. 1B) to illustrate interactions between life forms and their environments (Turner 2017). For Turner, cognitive plasticity embodies adaptations in system performance that are guided by both physiological (within-generation) and evolutionary (among-generation) feedbacks. In his framework, the boundaries of living systems (e.g., cell membranes) regulate exchanges of energy and materials rather than information (Turner 2009b). To Turner, living systems can access as much free energy as possible by building their own niches, moving within their environments, or modifying their phenotypes to better fit their environments (Turner 2009a).
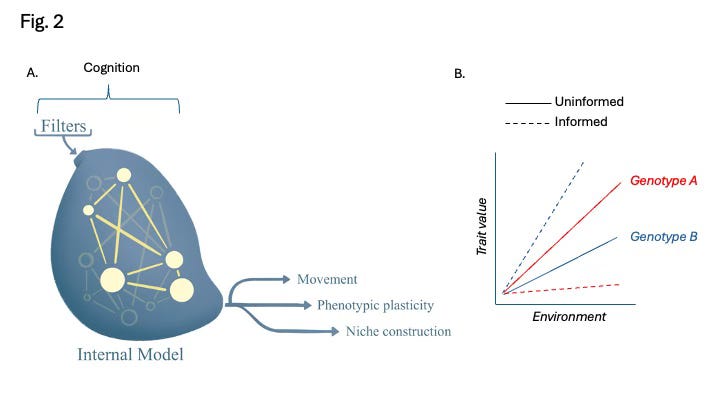
Both Friston’s and Turner’s models reference free energy as the disparity between a system’s current state and a reference state, though Friston emphasizes informational differences while Turner focuses on chemical or energetic conditions (Fig. 1). Both envision non-equilibrium systems encased by barriers allowing bidirectional flows, with systems continually updating themselves in response to new environmental information. Crucially, both models also emphasize reciprocal causality (Svensson 2018, Mitchell 2023): that systems shape their environments, which in turn shape future system responses. Fig. 2 depicts critical aspects of such systems.
An empirical roadmap to studying cognitive plasticity
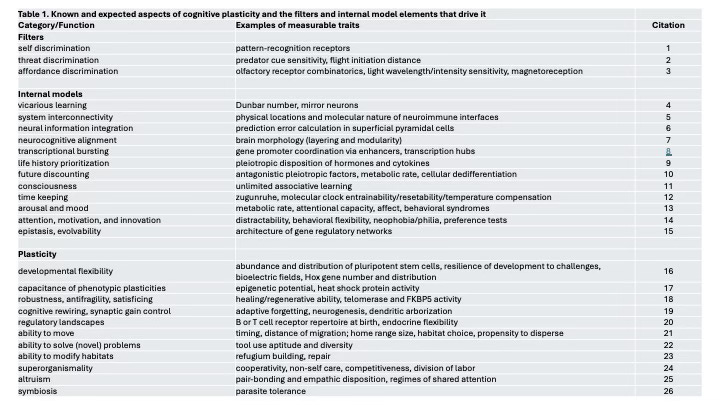
What about we empiricists? How can we contribute to a better understanding of the role of cognitive plasticity in biology? Friston’s and Turner’s conceptual models of living systems identify three aspects of cognitive plasticity that warrant attention: filters that determine what information is made available to internal models, internal models where decisions are made and responsiveness to information updated, and actions, the forms of plasticity that change the world or the system’s form within it (Fig. 2A). Many of these factors are very well-studied already (Table 1), but with some exceptions, they have not been considered as components of cognitive plasticity.
Filters, specifically semiotic ones, ensure that systems obtain and act on ecologically and evolutionarily relevant information. Many such filters are already well-characterized, such as the vertebrate Toll-like receptors (TLRs), which bind and recognize molecular motifs of bacteria, fungi, and viruses (Medzhitov and Janeway 1998). Metazoans could in principle respond to a functionally infinite variety of molecules (Deboer and Perelson 1993, Tauber 2017), yet in practice only a small subset bind TLRs and induce immune responses. TLRs are thus semiotic because they discriminate relevant environmental information from noise. They thus necessarily encode information about infection risk as shaped by the evolutionary legacies of ancestors including parents (via maternal and epigenetic effects) (Adami 2024). Analogous relationships exist between photons and photoreceptors, volatiles and olfactory receptors, toxins and cytochromes, and even metazoan hormones, cytokines, growth factors and their receptors. Rarely, though, has intraspecific variation in these factors been considered in the light of cognitive plasticity. Indeed, we know quite a bit about how variation in the architecture of TLRs and other semiotic filters affects health, but the fitness consequences of variation in natural populations are poorly known. Notable exceptions include opsins (Shichida and Matsuyama 2009, Hagen et al. 2023) and olfactory receptors (Finnerty et al. 2022). Perhaps it is time to start asking, among many things: how different are the semiotic information streams perceived by different individuals within populations (i.e., how different are their umwelts?), and how does that variation map onto fitness? Is there a relationship between rate of acquisition of semiotic information, the nature of internal model updating, and plastic responses within individuals?
Some internal models are also well-studied, especially gene and neural regulatory networks (Shen-Orr et al. 2002, Wagner et al. 2007, Newman 2010, O’Connell and Hofmann 2012). However, even the basic structure of internal models at other levels of organization are under-described (e.g., physiological regulatory networks) (Cohen et al. 2012, Martin and Cohen 2014), much less the computational processes within them (Levin 2019) and linkages among them (Okasha 2006). Understanding internal models is important because they are the processes that underpin cognitive decisions (Bongard and Levin 2023), but they will be particularly difficult to study because they are dynamic over multiple spatiotemporal scales. Fortunately, there have been recent advances to study these models (Kriegman et al. 2020, Alon 2023) including methods directed to coarse-grain lower-level variables into analogs of hyperparameters, what some have called ‘slow variables’ (Flack 2017). Approaches like these have already helped us understand synaptic plasticity and neural architecture, which control higher-level traits such as learning rate and cognition (Dellaferrera et al. 2022). Key questions remain though. Do particular model states underpin health, disease, and other complex phenotypic states related to fitness (Alon 2023)? Are there attractors in the landscape of model states (Demicheli and Coradini 2011), and how do evolutionary and ecological forces shape them (Martin et al. 2016)? When do systems update their models versus act differently in/on the world?
Examples of cognitive plasticity
To make the above abstractions as actionable as possible, I offer three concrete aspects of cognitive plasticity that could be investigated fairly easily and cost-effectively in many taxa. Critically, they are not nearly the only things that underpin cognitive plasticity (Table 1); they are simply illustrations. The first is epigenetic potential, which describes how cells use (semiotic) information and molecular epigenetic processes such as DNA methylation to match the phenotype to current and historical contexts (Kilvitis et al. 2017). In many species, CpG motifs (i.e., cytosines preceding guanines in a DNA sequence) can be methylated or unmethylated to influence gene expression (Jaenisch and Bird 2003). In vertebrates, many methyl marks are laid down over development, never to change, a process that contributes in part to cell differentiation. Other methyl marks are labile, being added or removed in response to changes in diet, hormones, methyltransferase activity, and other environmental factors (Vogt 2021).
An example of epigenetic potential with more direct connections to cognitive plasticity involves my work on one of the most broadly distributed animals on Earth, the house sparrow (Passer domesticus). In several studies, students, colleagues and I revealed epigenetic potential as important for the geographic range expansion of this species (Schrey et al. 2012, Schrey et al. 2014, Sheldon et al. 2018, Hanson et al. 2022). In one study in particular, house sparrows in newly-founded populations had more epigenetic potential (i.e., more CpG than GpC motifs) across their genomes than did birds from long-colonized sites (Hanson et al. 2022). This particular pattern seems to have arisen via natural selection, perhaps because individual sparrows with high epigenetic potential were better protected against pathogen infection than birds with low epigenetic potential, as our experimental data have shown (Sheldon et al. 2023). From a cognitive plasticity perspective, high epigenetic potential could represent a comparatively large repository of latent, cognitive plasticity for a colonizing bird (Kilvitis et al. 2017, Walsh and Sultan 2024), being adaptive because the genome of a bird first arriving in new areas would better poised to learn from and act on local information about disease risk, mitigate transposon activity (Marin et al. 2020), or both. In general, successful invaders seem to be those that are most behaviorally flexible (Sol et al. 2002). Perhaps the same holds for cognitive plasticity in immune gene expression.
Epigenetic potential relates to the ability of living systems to update their internal models, but endocrine flexibilityrepresents an example of the set of phenotypic options (i.e., actions) realizable by a cognitive system (Martin and Zimmer 2022, Zimmer et al. 2020, Zimmer et al. 2024, Zimmer et al. 2022). In other words, endocrine flexibility describes the landscape of regulated hormone variation available to an individual, with release contingent on the information the system encounters (i.e., how stressful it is). Flexibility in the regulation of many hormones warrants study, but one of the most pleiotropic steroid groups to consider is the glucocorticoids (Romero and Wingfield 2015). Glucocorticoids regulate glucose metabolism, but they also help vertebrates avoid, endure, or recover from physical and psychological stressors. Almost since their discovery, glucocorticoids have been studied by measuring their circulating concentrations and relating variation in concentrations among or within-individual animals to health or fitness (Bonier et al. 2009, Breuner et al. 2008). Whereas this approach has been productive, others (Taff and Vitousek 2016) and I think that the study of endocrine flexibility could help rectify these shortcomings because it captures the set of plastic responses latent in an individual (Baldan et al. 2021, Bonier and Martin 2016), not just generic phenotypic variation. To date, we have implicitly assumed there is one best endocrine solution to all problems, but endocrine flexibility recognizes that trait under selection might be the disposition to decide and mobilize the best hormonal response given the challenge (Zimmer et al. 2022), not mount the one best response, not one particular variant (Walsh and Sultan 2024).
A final measurable example of cognitive plasticity comes from work on insect nutrient regulation. Insect herbivores can be remarkably adept at obtaining the macronutrients they need to grow and reproduce while exploiting or avoiding plant secondary chemistry and minimizing risks from predators and parasitoids (Behmer 2009). A major step forward in understanding nutrient regulation occurred thirty years ago, with the development of the geometric framework (Raubenheimer and Simpson 1993, Simpson and Raubenheimer 1993). Using that framework, one can resolve how insects balance the intake of multiple nutrients simultaneously to meet their physiological needs. The proportions of nutrients (e.g., proteins, carbohydrates, lipids) in each diet are represented as axes in space, and insects move through that space by consuming different available foods at different rates. This approach provides a natural way to understand insect feeding goals, as most insects have well-defined intake targets and that also support high rates of growth. In terms of cognitive plasticity, one could view intake targets as internal models that insects use to provision their tissues with optimal nutrition using a broad variety of forms of information including the physiology of taste (Kvello 2024), post-ingestive regulation (Behmer 2009), and learning (Dukas and Bernays 2000). Other studies have examined how physiological and behavioral approaches to nutrient regulation change when insects are challenged with plant secondary compounds (i.e., when nutritionally superior foods also are protected by higher levels of deterrents or toxins) or predators and parasitoids (i.e., when consuming some foods carry additional risks of mortality). The geometric framework thus provides a way to visualize the state of internal models and is thus a rich experimental paradigm for analyzing how behavioral, physiological, and ecological forces dynamically alter those models. Generally, a similar framework could apply to many physiological tradeoffs, phenomena so important to the development of evolutionary theory (Stearns 1992, Garland Jr et al. 2022).
Looking forward
Reaction norms have been popular because they have worked so well for so many traits, and by work I mean effectively partition the causes of phenotypic variation into environmental or genetic sources. Here, I have questioned the reaction norm approach will work for cognitive plasticities, but I hope I’ve also identified elements important to the alternative framework necessitated by life as a dynamic process. Although reaction norms will not be appropriate for forms of cognitive plasticity themselves, it would be interesting to investigate whether they could be useful for particular facets of cognitive plasticity. For instance, one could describe reaction norms for attention, motivation, and a variety of other informational filters and internal models (Table 1) and probably learn important things about landscapes of latent plasticity. In this sense, we would not be modeling cognitive plasticity per se, but the scope of phenotypic options available to an organism should it encounter an environment (Wright 1931). In other words, maybe we can yet use the reaction norm to describe G, E, and GxE in the ability to be plastic, or plasticity in plasticity if you like. This condition seems to be what Wolterek (1909) meant reaction norms to capture anyway when he proposed them as ‘an organism’s multifaceted repertoire of contingent responses’ (Walsh and Sultan 2024).
I think that work on cognitive plasticity, generally, will give us a new appreciation about robustness, resilience and even antifragility from molecular to ecosystem levels. Resilience and robustness imply returns to baselines, and whereas for many plastic traits, this perspective is helpful and valid, for others, it does not apply. For cognitive traits, the return-to-safety mindset misses that many if not most plastic responses evolved to improve and perhaps become more adaptive through time. I expect that investigations of such biological antifragility will be of great value to conservation and medicine. Whereas homeostasis, allostasis and related concepts have been among the most powerful biological concepts ever offered, they are somewhat misleading for many processes (i.e., compensatory growth, neurogenesis, hormesis, most immune responses).
Finally, I close by noting that cognitive plasticity links naturally to the idea of agency, the capacity of a system to ‘understand,’ ‘make decisions,’ and ‘take action’. All living systems – across all branches of life and all levels – use information, are cognitive, and thus must have some level of agency (Moczek and Sultan 2023, Fábregas-Tejeda et al. 2024, Walsh 2015). Agency is presently anathema to many in biology, but it is the sole concept that unifies information with the other physical phenomena inherent to life (i.e., energy and matter) (Friston 2010, Ramstead et al. 2018, Hesp et al. 2019, Friston et al. 2023, Mitchell 2023). It is also a very old idea, tracing back to Aristotle and Epicurus, who emphasized that living things (humans, in their case) do things whereas non-living things have things done to them (Walsh 2015, Levin and Dennett 2020). In 1863, Thomas Reid went farther, arguing for “agent causation,’ that living systems have causal power not entirely reducible to the causal power of their component parts (Potter and Mitchell 2022). In modern times, almost all biologists have come to appreciate that living systems modify themselves such that non-genetic, phenotypic variation can become subject to natural selection (as in the Baldwin effect (Loison 2019, Baldwin 2018). For decades, we have also known that organisms choose when and where to move or modify their environments, both of which alters the speed and direction of evolution of their traits (as in the Bogert effect) (Huey et al. 2003, Pincebourde and Casas 2019, Muñoz 2022). I think that the main reason we have not used the word agency to describe these phenomena (besides the religious connotations of the word) is that it was not very clear that it would matter if we did so (Potter and Mitchell 2024). I do not claim here to have provided such specific guidance, but I hope that this paper inspires efforts to infuse our mathematical models of life with the key processes of life, replacing the dominant paradigm of change via accident and selection (Ågren 2021) with a paradigm that gives equal weight to how life uses information to remain far from thermodynamic equilibrium (Mitchell 2023, Walsh 2015, Davies 2019, Ramstead et al. 2018). I think that agency, like no other biological concept, explains both how life evolves and how it operates.
Acknowledgements:
I thank Art Woods and Cameron Ghalambor for help drafting earlier versions of the paper, several careful reads of other drafts, extensive feedback thereafter, years of constructive criticism about the ideas, and the friendship that enabled oftentimes energetic debates about material in the manuscript, all of which improved its final form. I also thank Mike Levin, Karl Friston, Scott Turner, Phil Ball, Sonia Sultan, Paul Davies, Denis Walsh, Erik Svensson, Sara Walker, Kevin Mitchell, Dan Nicholson and the many other guests of the Big Biology podcast that inspired the paper, refined my thinking, challenged my ideas, and filled gaps in our knowledge.
I also thank Ruth Demree, Brad van Paridon, Clayton Glasgow, and Molly Magid for helping transform those conversations into podcast episodes, which enabled many other colleagues to offer us critical and supportive feedback, too. Finally, I thank Keating Shahmehri for the images that visually enlivened the message in this paper, and Vania Assis, Kailey McCain, Nate Dowling, Diego Alarcon, Gabi Cifarelli, Dave Westneat, Zac Cheviron, Beau Larkin, and Matt Venesky for thoughtful feedback on earlier drafts, and Aaron Schrey, Haley Hanson, and Cedric Zimmer for many conversations that improved parts of the paper. I also recognize grants 0920475, 2027040, 2110070, and 2110233 from the National Science Foundation for support while writing the manuscript and for support to collect the data that bolster the ideas in the paper.
Glossary
Affordances. Opportunities in local environments that arise by virtue of a system interacting with that environment. The idea of affordances recognizes that what is available in a particular place or time is not simply imposed by the environment but rather emerges from an interaction between a complex set of potentials that are realized by the living system itself via its filters, internal model, and actions.
Agency. The capacity of a living system to act intentionally in response to its environment. It implies the ability to acquire information, make decisions, and exert control over its actions across multiple time scales.
Antifragile. Biological systems and forms of plasticity that improve functionally after exposure to challenges.
Cognitive. Used broadly to refer to the ability of complex systems to acquire information about their environment and to process that information to decide about how to respond. Cognition does not require a brain, nor even a nervous system.
Endocrine flexibility. The collective of endocrine responses available to an individual; latent endocrine variation to be released contingent on a challenge and after cognitive processing.
Epigenetic potential. The ability of whole genomes or gene regulatory regions to use epigenetic processes to learn. One example involves the number and distribution of CpG motifs in gene regulatory regions, especially promoters, that can be methylated or unmethylated.
Filter. Part of a non-equilibrium steady-state system that constrains the kinds of information available to the system’s internal models. A key mechanism by which internal models discriminate semiotic information from noise.
Free energy. Evaluating how closely incoming information matches what is expected by an internal model by measuring ‘surprise’. In this context, the term ‘free energy’ was developed by Karl Friston by analogy with information theory and entropy.
Information. Used in two senses in this paper. Information can be syntactic, as defined by Claude Shannon and used in information theory. This use focuses on the statistical structure of messages, the number of bytes required to send them, and the sources of noise that can degrade information as it passes through different channels. Information can also be semiotic, which focuses more on its meaning; here, the difference that makes a functional difference.
Internal model. A network of interacting set of biological parts (e.g., genes, proteins, neurons, tissues, etc.) that processes incoming information, computes what that information reveals about internal and external states, compares those states to the model’s expected state, and effects downstream changes on the collective system or the world.
Markov blanket. Informational boundaries around complex systems that act as the interface between a system and its surrounding environment. This boundary enables the system to remain viable but also independent from the environment.
Niche construction. The process by which organisms alter their local environments. These alterations can affect the way organisms interact with resources and other entities as well as the nature and magnitude of selection on populations.
References
ADAMI, C. 2024. The Evolution of Biological Information: How Evolution Creates Complexity, from Viruses to Brains, Princeton University Press.
ÅGREN, J. A. 2021. The gene’s-eye view of evolution, Oxford University Press.
ALON, U. 2023. Systems medicine: physiological circuits and the dynamics of disease, CRC Press.
ARAYA-AJOY, Y. G. and DINGEMANSE, N. J. 2014. Characterizing behavioural ‘characters’: an evolutionary framework. -Proceedings of the Royal Society B: Biological Sciences, 281: 20132645.
ARNOLD, S. J. 1983. Morphology, Performance and Fitness. -American Zoologist, 23: 347-361.
BALDAN, D., NEGASH, M. and OUYANG, J. Q. 2021. Are individuals consistent? Endocrine reaction norms under different ecological challenges. -Journal of Experimental Biology, 224.
BALDWIN, J. M. 2018. A new factor in evolution. -Diacronia: 1-13.
BEHMER, S. T. 2009. Insect herbivore nutrient regulation. -Annual review of entomology, 54: 165-187.
BERNHARDT, J. R., O’CONNOR, M. I., SUNDAY, J. M. and GONZALEZ, A. 2020. Life in fluctuating environments. -Philosophical Transactions of the Royal Society B, 375: 20190454.
BONGARD, J. and LEVIN, M. 2023. There’s plenty of room right here: Biological systems as evolved, overloaded, multi-scale machines. -Biomimetics, 8: 110.
BONIER, F. and MARTIN, P. R. 2016. How can we estimate natural selection on endocrine traits? Lessons from evolutionary biology. Proc. R. Soc. B. The Royal Society, pp. 20161887.
BONIER, F., MARTIN, P. R., MOORE, I. T. and WINGFIELD, J. C. 2009. Do baseline glucocorticoids predict fitness? -Trends in Ecology & Evolution, 24: 634-642.
BREUNER, C. W., PATTERSON, S. H. and HAHN, T. P. 2008. In search of relationships between the acute adrenocortical response and fitness. -General and Comparative Endocrinology, 157: 288-295.
COHEN, A. A., MARTIN, L. B., WINGFIELD, J. C., MCWILLIAMS, S. R. and DUNNE, J. A. 2012. Physiological regulatory networks: ecological roles and evolutionary constraints. -Trends in Ecology & Evolution, 27: 428-435.
COLOMBO, M. and WRIGHT, C. 2021. First principles in the life sciences: the free-energy principle, organicism, and mechanism. -Synthese, 198: 3463-3488.
DANTZER, B. 2023. Frank Beach Award Winner: The centrality of the hypothalamic-pituitary-adrenal axis in dealing with environmental change across temporal scales. -Hormones and Behavior, 150.
DAVIES, P. 2019. The Demon in the machine: How hidden webs of information are solving the mystery of life, University of Chicago Press.
DEBOER, R. J. and PERELSON, A. S. 1993. How Diverse Should the Immune-System Be. -Proceedings of the Royal Society of London Series B-Biological Sciences, 252: 171-175.
DEL GIUDICE, M., BUCK, C. L., CHABY, L. E., GORMALLY, B. M., TAFF, C. C., THAWLEY, C. J., VITOUSEK, M. N. and WADA, H. 2018. What is stress? A systems perspective. -Integrative and comparative biology, 58: 1019-1032.
DELLAFERRERA, G., WOźNIAK, S., INDIVERI, G., PANTAZI, A. and ELEFTHERIOU, E. 2022. Introducing principles of synaptic integration in the optimization of deep neural networks. -Nature Communications, 13: 1885.
DEMICHELI, R. and CORADINI, D. 2011. Gene regulatory networks: a new conceptual framework to analyse breast cancer behaviour. -Annals of Oncology, 22: 1259-1265.
DINGEMANSE, N. J., KAZEM, A. J. N., REALE, D. and WRIGHT, J. 2010. Behavioural reaction norms: animal personality meets individual plasticity. -Trends in Ecology & Evolution, 25: 81-89.
DINGEMANSE, N. J. and WOLF, M. 2013. Between-individual differences in behavioural plasticity within populations: causes and consequences. -Animal Behaviour, 85: 1031-1039.
DUKAS, R. and BERNAYS, E. A. 2000. Learning improves growth rate in grasshoppers. -Proceedings of the National Academy of Sciences, 97: 2637-2640.
DUPONT, L., THIERRY, M., ZINGER, L., LEGRAND, D. and JACOB, S. 2024. Beyond reaction norms: the temporal dynamics of phenotypic plasticity. -Trends in Ecology & Evolution, 39: 41-51.
FáBREGAS-TEJEDA, A., BAEDKE, J., PRIETO, G. I. and RADICK, G. 2024. The riddle of organismal agency: New historical and philosophical reflections, Taylor & Francis.
FEINER, N., FELDMAN, M., GILBERT, S. F., LALA, K. N. and ULLER, T. 2024. Evolution evolving: The developmental origins of adaptation and biodiversity, Princeton University Press.
FINNERTY, P. B., MCARTHUR, C., BANKS, P., PRICE, C. and SHRADER, A. M. 2022. The olfactory landscape concept: a key source of past, present, and future information driving animal movement and decision-making. -BioScience, 72: 745-752.
FLACK, J. C. 2017. Coarse-graining as a downward causation mechanism. -Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences, 375: 20160338.
FRISTON, K. 2010. The free-energy principle: a unified brain theory? -Nature reviews neuroscience, 11: 127-138.
FRISTON, K., DA COSTA, L., SAJID, N., HEINS, C., UELTZHöFFER, K., PAVLIOTIS, G. A. and PARR, T. 2023. The free energy principle made simpler but not too simple. -Physics Reports, 1024: 1-29.
GARLAND JR, T., DOWNS, C. J. and IVES, A. R. 2022. Trade-offs (and constraints) in organismal biology. -Physiological and Biochemical Zoology, 95: 82-112.
GHALAMBOR, C. K., HOKE, K. L., RUELL, E. W., FISCHER, E. K., REZNICK, D. N. and HUGHES, K. A. 2015. Non-adaptive plasticity potentiates rapid adaptive evolution of gene expression in nature. -Nature, 525: 372-375.
GHALAMBOR, C. K., MCKAY, J. K., CARROLL, S. P. and REZNICK, D. N. 2007a. Adaptive versus non-adaptive phenotypic plasticity and the potential for contemporary adaptation in new environments. -Functional Ecology, 21: 394-407.
GHALAMBOR, C. K., MCKAY, J. K., CARROLL, S. P. and REZNICK, D. N. 2007b. Adaptive versus non‐adaptive phenotypic plasticity and the potential for contemporary adaptation in new environments. -Functional ecology, 21: 394-407.
GILBERT, S. F. and EPEL, D. 2009. Ecological developmental biology: integrating epigenetics, medicine, and evolution.
GLUCKMAN, P., BEEDLE, A. and HANSON, M. 2009. Principles of evolutionary medicine, Oxford University Press.
GOTTLIEB, G. 2002. Individual development and evolution, Mahwah, NJ, Lawrence Erlbaum Associates.
HAGEN, J. F., ROBERTS, N. S. and JOHNSTON JR, R. J. 2023. The evolutionary history and spectral tuning of vertebrate visual opsins. -Developmental biology, 493: 40-66.
HAIG, D. 1996. Placental hormones, genomic imprinting, and maternal-fetal communication. -Journal of Evolutionary Biology, 9: 357-380.
HANSON, H. E., WANG, C. Q., SCHREY, A. W., LIEBL, A. L., RAVINET, M., JIANG, R. H. Y. and MARTIN, L. B. 2022. Epigenetic Potential and DNA Methylation in an Ongoing House Sparrow (Passer domesticus) Range Expansion. -American Naturalist.
HAU, M., DEIMEL, C. and MOIRON, M. 2022. Great tits differ in glucocorticoid plasticity in response to spring temperature. -Proceedings of the Royal Society B-Biological Sciences, 289.
HEDRICK, S. M. 2004. The acquired immune system: A vantage from beneath. -Immunity, 21: 607-615.
HESP, C., RAMSTEAD, M., CONSTANT, A., BADCOCK, P., KIRCHHOFF, M. and FRISTON, K. 2019. A multi-scale view of the emergent complexity of life: A free-energy proposal. Evolution, development and complexity: multiscale evolutionary models of complex adaptive systems. Springer, pp. 195-227.
HOFSTADTER, D. R. 2007. I am a strange loop, Basic books.
HUEY, R. B., HERTZ, P. E. and SINERVO, B. 2003. Behavioral drive versus behavioral inertia in evolution: a null model approach. -The American Naturalist, 161: 357-366.
HUXLEY, J. 1942. Evolution. The modern synthesis.
JABLONKA, E., LAMB, M. J. and ZELIGOWSKI, A. 2014. Evolution in Four Dimensions, revised edition: Genetic, Epigenetic, Behavioral, and Symbolic Variation in the History of Life, MIT press.
JACOB, F. and MONOD, J. 1961. Genetic regulatory mechanisms in the synthesis of proteins. -Journal of molecular biology, 3: 318-356.
JAENISCH, R. and BIRD, A. 2003. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. -Nature Genetics, 33: 245-254.
KILVITIS, H. J., HANSON, H., SCHREY, A. W. and MARTIN, L. B. 2017. Epigenetic Potential as a Mechanism of Phenotypic Plasticity in Vertebrate Range Expansions. -Integrative and comparative biology, 57: 385-395.
KINGSOLVER, J. G., GOMULKIEWICZ, R. and CARTER, P. A. 2001. Variation, selection and evolution of function-valued traits. Microevolution Rate, Pattern, Process. Springer, pp. 87-104.
KIRCHHOFF, M., PARR, T., PALACIOS, E., FRISTON, K. and KIVERSTEIN, J. 2018. The Markov blankets of life: autonomy, active inference and the free energy principle. -Journal of The royal society interface, 15: 20170792.
KRIEGMAN, S., BLACKISTON, D., LEVIN, M. and BONGARD, J. 2020. A scalable pipeline for designing reconfigurable organisms. -Proceedings of the National Academy of Sciences, 117: 1853-1859.
KURTZ, J. and ARMITAGE, S. A. O. 2006. Alternative adaptive immunity in invertebrates. -Trends in Immunology, 27: 493-496.
KVELLO, P. 2024. Insect Taste. The Concept of Ecostacking: Techniques and Applications. CABI GB, pp. 79-93.
LALAND, K., ULLER, T., FELDMAN, M., STERELNY, K., MüLLER, G. B., MOCZEK, A., JABLONKA, E., ODLING-SMEE, J., WRAY, G. A. and HOEKSTRA, H. E. 2014. Does evolutionary theory need a rethink? -Nature, 514: 161-164.
LANDE, R. 2009. Adaptation to an extraordinary environment by evolution of phenotypic plasticity and genetic assimilation. -Journal of Evolutionary Biology, 22: 1435-1446.
LEDON-RETTIG, C. C., PFENNIG, D. W. and CRESPI, E. J. 2010. Diet and hormonal manipulation reveal cryptic genetic variation: implications for the evolution of novel feeding strategies. -Proceedings of the Royal Society B: Biological Sciences, 277: 3569-3578.
LEVIN, M. 2019. The computational boundary of a “self”: developmental bioelectricity drives multicellularity and scale-free cognition. -Frontiers in psychology, 10: 2688.
LEVIN, M. 2023. Darwin’s agential materials: evolutionary implications of multiscale competency in developmental biology. -Cellular and Molecular Life Sciences, 80: 142.
LEVIN, M. and DENNETT, D. C. 2020. Cognition all the way down. -Aeon Essays.
LEWONTIN, R. C. 1970. The units of selection. -Annual review of ecology and systematics: 1-18.
LOISON, L. 2019. Canalization and genetic assimilation: Reassessing the radicality of the Waddingtonian concept of inheritance of acquired characters. Seminars in cell & developmental biology. Elsevier, pp. 4-13.
LYON, P., KEIJZER, F., ARENDT, D. and LEVIN, M. 2021. Reframing cognition: getting down to biological basics. The Royal Society, pp. 20190750.
MARIN, P., GENITONI, J., BARLOY, D., MAURY, S., GIBERT, P., GHALAMBOR, C. K. and VIEIRA, C. 2020. Biological invasion: The influence of the hidden side of the (epi) genome. -Functional Ecology, 34: 385-400.
MARTIN, J. G. A., NUSSEY, D. H., WILSON, A. J. and RéALE, D. 2011. Measuring individual differences in reaction norms in field and experimental studies: a power analysis of random regression models. -Methods in Ecology and Evolution, 2: 362-374.
MARTIN, L. B., BURGAN, S. C., ADELMAN, J. S. and GERVASI, S. S. 2016. Host Competence: An Organismal Trait to Integrate Immunology and Epidemiology. -Integrative and Comparative Biology, 56: 1225-1237.
MARTIN, L. B. and COHEN, A. 2014. Physiological regulatory networks: the orchestra of life? In: MARTIN, L. B., GHALAMBOR, C. K. and WOODS, H. A. (eds.) Integrative Organismal Biology. Wiley.
MARTIN, L. B., HANSON, H. E., HAUBER, M. E. and GHALAMBOR, C. K. 2021. Genes, Environments, and Phenotypic Plasticity in Immunology. -Trends in Immunology.
MARTIN, L. B. and ZIMMER, C. 2022. Endocrine flexibility. -Journal of Experimental Biology, 225.
MCWILLIAMS, S. R. and KARASOV, W. H. 2014. Spare capacity and phenotypic flexibility in the digestive system of a migratory bird: defining the limits of animal design. -Proceedings. Biological Sciences, 281: 20140308.
MEDZHITOV, R. and JANEWAY, C. A., JR. 1998. An ancient system of host defense. -Curr Opin Immunol, 10: 12-5.
MITCHELL, K. J. 2023. Free agents: how evolution gave us free will.
MOCZEK, A. P., SULTAN, S., FOSTER, S., LEDóN-RETTIG, C., DWORKIN, I., NIJHOUT, H. F., ABOUHEIF, E. and PFENNIG, D. W. 2011. The role of developmental plasticity in evolutionary innovation. -Proceedings of the Royal Society B: Biological Sciences, 278: 2705-2713.
MOCZEK, A. P. and SULTAN, S. E. 2023. Agency in living systems. -Evolution & Development, 25.
MüLLER-HILL, B. and OEHLER, S. 1996. The lac operon.
MUñOZ, M. M. 2022. The Bogert effect, a factor in evolution. -Evolution, 76: 49-66.
NELSON, R. J., DEMAS, G. E., KLEIN, S. L. and KRIEGSFELD, L. J. 2002. Seasonal patterns of stress, immune function, and disease., New York, Cambridge University Press.
NEWMAN, S. A. 2010. Dynamic patterning modules. In: PIGLIUCCI, M. and MULLER, G. B. (eds.) Evolution: the Extended Synthesis. MIT Press, Cambridge, MA, pp. 281-306.
NICHOLSON, D. J. 2019. Is the cell really a machine? -Journal of theoretical biology, 477: 108-126.
NICHOLSON, D. J. and DUPRé, J. 2018. Everything flows: towards a processual philosophy of biology, Oxford University Press.
NOBLE, D. 2006. The Music of Life: biology beyond genes, New York, Oxford UP.
NOBLE, D. 2008. The music of life: biology beyond genes, Oxford University Press, USA.
NUSSEY, D. H., WILSON, A. J. and BROMMER, J. E. 2007. The evolutionary ecology of individual phenotypic plasticity in wild populations. -Journal of Evolutionary Biology, 20: 831-844.
O’CONNELL, L. A. and HOFMANN, H. A. 2012. Evolution of a vertebrate social decision-making network. -Science, 336: 1154-1157.
O’CONNOR, M. I., PENNELL, M. W., ALTERMATT, F., MATTHEWS, B., MELIáN, C. J. and GONZALEZ, A. 2019. Principles of ecology revisited: Integrating information and ecological theories for a more unified science. -Frontiers in Ecology and Evolution, 7: 219.
ODLING-SMEE, F. J., LALAND, K. N. and FELDMAN, M. W. 1996. Niche construction. -The American Naturalist, 147: 641-648.
OKASHA, S. 2006. Evolution and the levels of selection, Clarendon Press.
OKASHA, S. 2018. Agents and goals in evolution, Oxford University Press.
PIERSMA, T. and DRENT, J. 2003. Phenotypic flexibility and the evolution of organismal design. -Trends in Ecology & Evolution, 18: 228-233.
PIGLIUCCI, M. 2007. Do we need an extended evolutionary synthesis? -Evolution, 61: 2743-2749.
PIGLIUCCI, M. 2010. Genotype-phenotype mapping and the end of the ‘genes as blueprint’ metaphor. -Philosophical Transactions of the Royal Society B-Biological Sciences, 365: 557-566.
PIGLIUCCI, M. and MüLLER, G. B. 2010. Elements of an extended evolutionary synthesis. -Evolution: The extended synthesis: 3-17.
PINCEBOURDE, S. and CASAS, J. 2019. Narrow safety margin in the phyllosphere during thermal extremes. -Proceedings of the National Academy of Sciences, 116: 5588-5596.
POTTER, H. D. and MITCHELL, K. J. 2022. Naturalising agent causation. -Entropy, 24: 472.
POTTER, H. D. and MITCHELL, K. J. 2024. A critique of the agential stance in development and evolution. The riddle of organismal agency. Routledge, pp. 131-149.
PRICE, T. D., QVARNSTROM, A. and IRWIN, D. E. 2003. The role of phenotypic plasticity in driving genetic evolution. -Proceedings of the Royal Society B, 270: 1433-1440.
RAMSTEAD, M. J. D., BADCOCK, P. B. and FRISTON, K. J. 2018. Answering Schrödinger’s question: A free-energy formulation. -Physics of life reviews, 24: 1-16.
RAUBENHEIMER, D. and SIMPSON, S. J. 1993. The geometry of compensatory feeding in the locust. -Animal behaviour, 45: 953-964.
RIVERA, H. E., AICHELMAN, H. E., FIFER, J. E., KRIEFALL, N. G., WUITCHIK, D. M., SMITH, S. J. and DAVIES, S. W. 2021. A framework for understanding gene expression plasticity and its influence on stress tolerance. -Molecular ecology, 30: 1381-1397.
ROMERO, L. M. and WINGFIELD, J. C. 2015. Tempests, poxes, predators, and people: stress in wild animals and how they cope, Oxford University Press.
SCHLICHTING, C. D. and PIGLIUCCI, M. 1998. Phenotypic evolution: a reaction norm perspective, Sinauer Associates Incorporated.
SCHMALHAUSEN, I. I. 1949. Factors of evolution: the theory of stabilizing selection.
SCHREY, A. W., COON, C. A. C., GRISPO, M. T., AWAD, M., IMBOMA, T., MCCOY, E. D., MUSHINSKY, H. R., RICHARDS, C. L. and MARTIN, L. B. 2012. Epigenetic Variation May Compensate for Decreased Genetic Variation with Introductions: A Case Study Using House Sparrows (Passer domesticus) on Two Continents. -Genetics Research International, 2012: 7.
SCHREY, A. W., LIEBL, A. L., RICHARDS, C. L. and MARTIN, L. B. 2014. Range Expansion of House Sparrows (Passer domesticus) in Kenya: Evidence of Genetic Admixture and Human-Mediated Dispersal. -Journal of Heredity, 105: 60-69.
SHAPIRO, J. A. 2011. Evolution: a view from the 21st century, Pearson education.
SHELDON, E., SCHREY, A., ANDREW, S., RAGSDALE, A. and GRIFFITH, S. 2018. Epigenetic and genetic variation among three separate introductions of the house sparrow (Passer domesticus) into Australia. -Royal Society open science, 5: 172185.
SHELDON, E., ZIMMER, C., HANSON, H., KOUSSAYER, B., SCHREY, A., REESE, D., WIGLEY, P., WEDLEY, A. L. and MARTIN, L. B. 2023. High epigenetic potential protects songbirds against pathogenic Salmonella enterica infection. -Journal of Experimental Biology, 226: jeb245475.
SHEN-ORR, S. S., MILO, R., MANGAN, S. and ALON, U. 2002. Network motifs in the transcriptional regulation network of Escherichia coli. -Nature genetics, 31: 64-68.
SHICHIDA, Y. and MATSUYAMA, T. 2009. Evolution of opsins and phototransduction. -Philosophical Transactions of the Royal Society B: Biological Sciences, 364: 2881-2895.
SIMPSON, S. J. and RAUBENHEIMER, D. 1993. A multi-level analysis of feeding behaviour: the geometry of nutritional decisions. -Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences, 342: 381-402.
SMALLEGANGE, I. M. 2022. Integrating developmental plasticity into eco-evolutionary population dynamics. -Trends in Ecology & Evolution, 37: 129-137.
SNELL-ROOD, E. C. 2013. An overview of the evolutionary causes and consequences of behavioural plasticity. -Animal Behaviour, 85: 1004-1011.
SNELL-ROOD, E. C. and EHLMAN, S. M. 2021. Ecology and evolution of plasticity. -Phenotypic plasticity & evolution: 139-160.
SOL, D., TIMMERMANS, S. and LEFEBVRE, L. 2002. Behavioural flexibility and invasion success in birds. -Animal Behaviour, 63: 495-502.
STEARNS, S. C. 1992. The Evolution of Life Histories, Oxford University Press.
SULTAN, S. E. 2019. Genotype-environment interaction and the unscripted reaction norm.
SVENSSON, E. I. 2018. On reciprocal causation in the evolutionary process. -Evolutionary Biology, 45: 1-14.
SVENSSON, E. I., GOMEZ-LLANO, M. and WALLER, J. T. 2020. Selection on phenotypic plasticity favors thermal canalization. -Proceedings of the National Academy of Sciences, 117: 29767-29774.
SWANSON, D. L. and MERKORD, C. 2013. Seasonal phenotypic flexibility of flight muscle size in small birds: a comparison of ultrasonography and tissue mass measurements. -Journal of Ornithology, 154: 119-127.
TAFF, C. C. and VITOUSEK, M. N. 2016. Endocrine Flexibility: Optimizing Phenotypes in a Dynamic World? -Trends in Ecology & Evolution, 31: 476-488.
TALEB, N. N. 2014. Antifragile: Things that gain from disorder, Random House Trade Paperbacks.
TAUBER, A. I. 2017. Immunity: the evolution of an idea, Oxford University Press.
TROTTER, J. H., LIEBL, A. L., WEEBER, E. J. and MARTIN, L. B. 2011. Linking ecological immunology and evolutionary medicine: the case for apolipoprotein E. -Functional Ecology, 25: 40-47.
TURNER, J. S. 2009a. The extended organism: the physiology of animal-built structures, Harvard University Press.
TURNER, J. S. 2009b. The tinkerer’s accomplice: how design emerges from life itself, Harvard University Press.
TURNER, J. S. 2016. Homeostasis and the physiological dimension of niche construction theory in ecology and evolution. -Evolutionary ecology, 30: 203-219.
TURNER, J. S. 2017. Purpose and desire: what makes something” alive” and why modern Darwinism has failed to explain it, HarperOne New York.
UVAROV, B.-P. 1921. A revision of the genus Locusta, L.(= Pachytylus, Fieb.), with a new theory as to the periodicity and migrations of locusts. -Bulletin of entomological Research, 12: 135-163.
VANCHURIN, V., WOLF, Y. I., KATSNELSON, M. I. and KOONIN, E. V. 2022. Toward a theory of evolution as multilevel learning. -Proceedings of the National Academy of Sciences, 119: e2120037119.
VIA, S., GOMULKIEWICZ, R., DEJONG, G., SCHEINER, S. M., SCHLICHTING, C. D. and VANTIENDEREN, P. H. 1995. Adaptive Phenotypic Plasticity - Consensus and Controversy. -Trends in Ecology & Evolution, 10: 212-217.
VOGT, G. 2021. Epigenetic variation in animal populations: sources, extent, phenotypic implications, and ecological and evolutionary relevance. -Journal of Biosciences, 46: 24.
VON BERTALANFFY, L. 1952. Problems of life; an evaluation of modern biological thought.
WADDINGTON, C. 2014. The strategy of the genes, Routledge.
WADDINGTON, C. H. 1953. Genetic assimilation of an acquired character. -Evolution: 118-126.
WADE, M. J. and SULTAN, S. E. 2023. Niche construction and the environmental term of the price equation: How natural selection changes when organisms alter their environments. -Evolution & Development, 25: 451-469.
WAGNER, G. N. P., PAVLICEV, M. and CHEVERUD, J. M. 2007. The road to modularity. -Nature Reviews Genetics, 8: 921-931.
WALKER, S. I., DAVIES, P. C. and ELLIS, G. F. 2017. From matter to life: information and causality, Cambridge University Press.
WALKER, S. I., KIM, H. and DAVIES, P. C. 2016. The informational architecture of the cell. -Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences, 374: 20150057.
WALSH, D. M. 2015. Organisms, agency, and evolution, Cambridge University Press.
WALSH, D. M. and SULTAN, S. E. 2024. The Higher-Order Norm of Reaction: Biological Agency and Adaptive Phenotypic Response. The Riddle of Organismal Agency. Routledge, pp. 114-130.
WATSON, R. A. and SZATHMáRY, E. 2016. How can evolution learn? -Trends in ecology & evolution, 31: 147-157.
WEST-EBERHARD, M. 2003. Developmental Plasticity and Evolution, Oxford, Oxford University Press.
WESTNEAT, D. F., HATCH, M. I., WETZEL, D. P. and ENSMINGER, A. L. 2011. Individual Variation in Parental Care Reaction Norms: Integration of Personality and Plasticity. -The American Naturalist, 178: 652-667.
WESTNEAT, D. F., POTTS, L. J., SASSER, K. L. and SHAFFER, J. D. 2019. Causes and consequences of phenotypic plasticity in complex environments. -Trends in Ecology & Evolution, 34: 555-568.
WILLIAMS, T. D. 2008. Individual variation in endocrine systems: moving beyond the ‘tyranny of the Golden Mean’. -Philosophical Transactions of the Royal Society B-Biological Sciences, 363: 1687-1698.
WOESE, C. R. 2004. A new biology for a new century. -Microbiology and molecular biology reviews, 68: 173-186.
WOLTERECK, R. 1909. Weitere experimentelle Untersuchungen uber Artveranderung, speziell uberdas Wesen quantitativer Artunterschyiede bei Daphniden. -Verh. D. Tsch. Zool. Ges, 1909: 110-172.
WOODS, H. A. and WILSON, J. K. 2014. An elephant in the fog: unifying concepts of physiological stasis and change. In: MARTIN, L. B., GHALAMBOR, C. K. and WOODS, H. A. (eds.) Integrative Organismal Biology. Wiley.
WRIGHT, J., HAALAND, T. R., DINGEMANSE, N. J. and WESTNEAT, D. F. 2022. A reaction norm framework for the evolution of learning: how cumulative experience shapes phenotypic plasticity. -Biological Reviews, 97: 1999-2021.
WRIGHT, S. 1931. Evolution in Mendelian populations. -Genetics, 16: 97.
ZIMMER, C., HANSON, H. E., WILDMAN, D. E., UDDIN, M. and MARTIN, L. B. 2020. FKBP5 : A Key Mediator of How Vertebrates Flexibly Cope with Adversity. -BioScience, 70: 1127-1138.
ZIMMER, C., JIMENO, B. and MARTIN, L. B. 2024. HPA flexibility and FKBP5: promising physiological targets for conservation. -Philosophical Transactions of the Royal Society B, 379: 20220512.
ZIMMER, C., WOODS, H. A. and MARTIN, L. B. 2022. Information theory in vertebrate stress physiology. -Trends in Endocrinology and Metabolism, 33: 8-17.